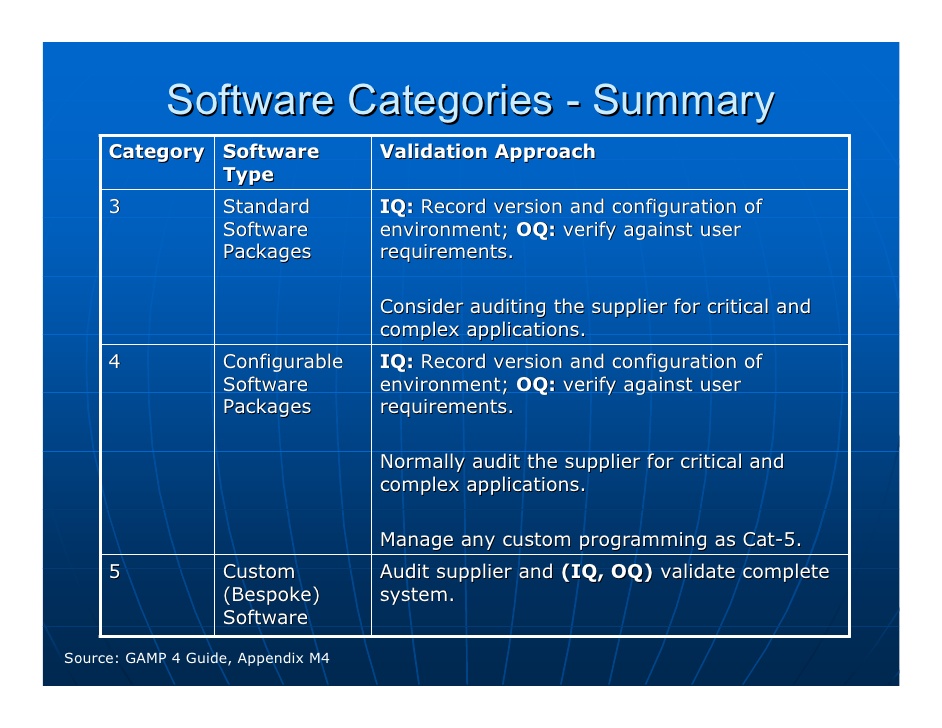
These changes include įDA cGMPs for the 21st Century initiative and associated guidance promoting science-based risk Why GAMP 5 Now?Since the release of GAMP 4 in 2001 the regulatory bodies had made significant updates in their thinking and approach to regulatory compliance. The GAMP guidances are accepted by regulators world wide and referenced by the FDA and PIC/S in their documents. Today GAMP is a global organization which has “communities of practice” (COP) in Europe, Japan, and the Americas. GAMP 4, which was released in 2001, has been one of the standards GxP companies have been using for computerized system validation for the last seven years. They joined forces with ISPE (International Society for Pharmaceutical Engineering) as a “technical sub-committee” and in 1995 released the first GAMP guidelines. GAMP HistoryGAMP (Good Automated Manufacturing Practice) was started in 1991 by a group of pharmaceutical experts in the UK who wanted to meet the changing FDA expectations for GMP compliance of manufacturing and related systems. 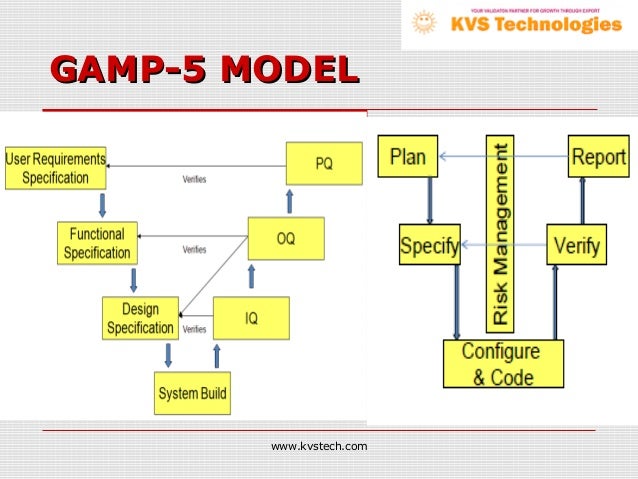
The purpose of the guidelines is to “provide a cost effective framework of good practice to ensure that computerized systems are fit for use and compliant with regulation.” 1 These guidelines are the latest, up-to-date thinking in the approach to validation of GxP computerized systems. The new GAMP®5 guidelines were released February 2008 at the ISPE Manufacturing Excellence Conference in Tampa, Florida. 2GAMP®5: A Risk-based Approach to Compliant GxP Computerized Systems
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
